 Historically, krypton was widely considered to be chemically inert however, since the 1960’s, studies have found several compounds of krypton – including krypton diflouride and an oxyacid salt containing krypton. Krypton is most often combined with other rare gases to make fluorescent lamps. Compare the configurations of neon and aluminum: Notation He Ne Ar Kr Xe Rn Ne (10 electrons) A1 (13 electrons) 3s2 3p1 The description of the. The element’s gas concentration in earth’s atmosphere is about 1 ppm and is isolated by fractioning liquid air. Electron Configuration and Oxidation States of Krypton Electron configuration of Krypton is Ar 3d10 4s2 4p6. The vibrant green and orange spectral-signatured element is considered a noble gas due to its low chemical reactivity. Because its boiling point (153.4 ☌, or 244. Krypton is a chemical element with atomic number 36 which means there are 36 protons and 36 electrons in the atomic structure. The element was found in residue of the evaporated components of liquid air by W. 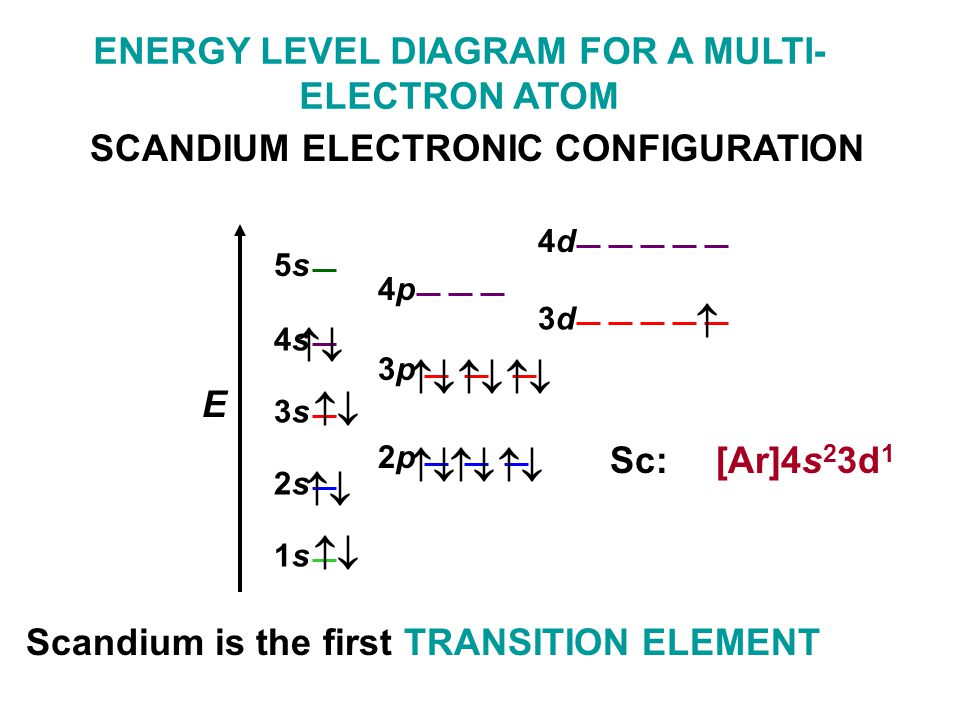
This indicates that it has a completely filled outer shell, which makes it chemically stable. A) Kr4s23d8 B) Ar4s4 C) Kr4s43d8 D) Ar3d104s24p6 E) Ar4s43d4 A) down a group and from right to left across a period Atomic radius generally increases as we move. The electron shell configuration of krypton is 2, 8, 18, 8. 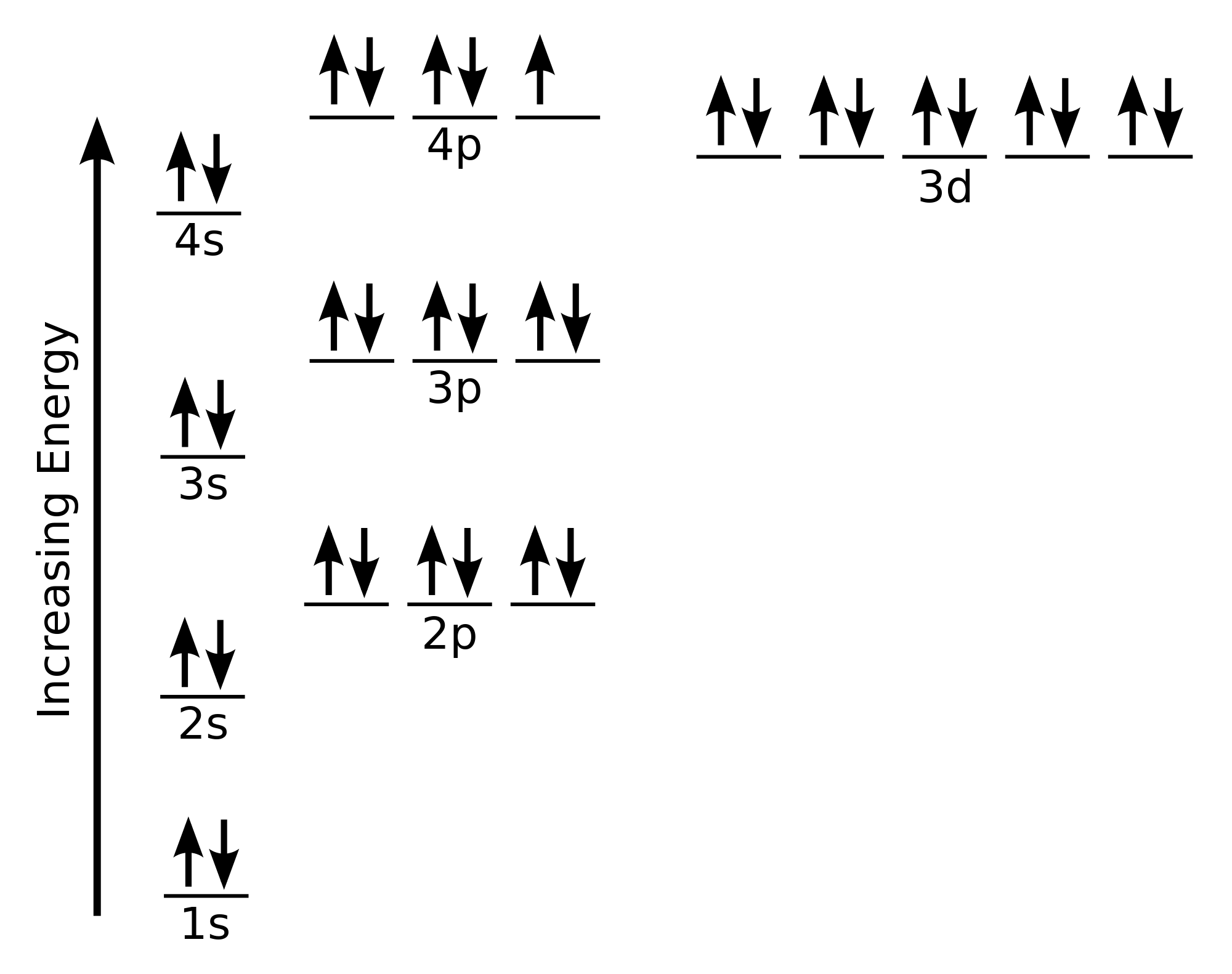
Krypton, named after the Greek word kryptos, meaning “hidden”, was discovered in 1898. Atomic Properties of Krypton Oxidation states: 2, 1, 0 Valence Electrons: 4s2 4p The ionization potential of an atom: 13.94 Ionization energies: 1st: 1350.8. Electron Configuration of the elements Krypton, Ar4s23d104p Tin, Kr5s24d105p Lanthanum, Xe6s25d Titanium, Ar4s23d Lawrencium, Rn7s25f147p. The condensed electron configuration of krypton, element 36, is. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
